An atom is the smallest part of an element that can exist. It consists of a dense, positively charged nucleus surrounded by negatively charged electrons that inhabit specific energy levels or shells.
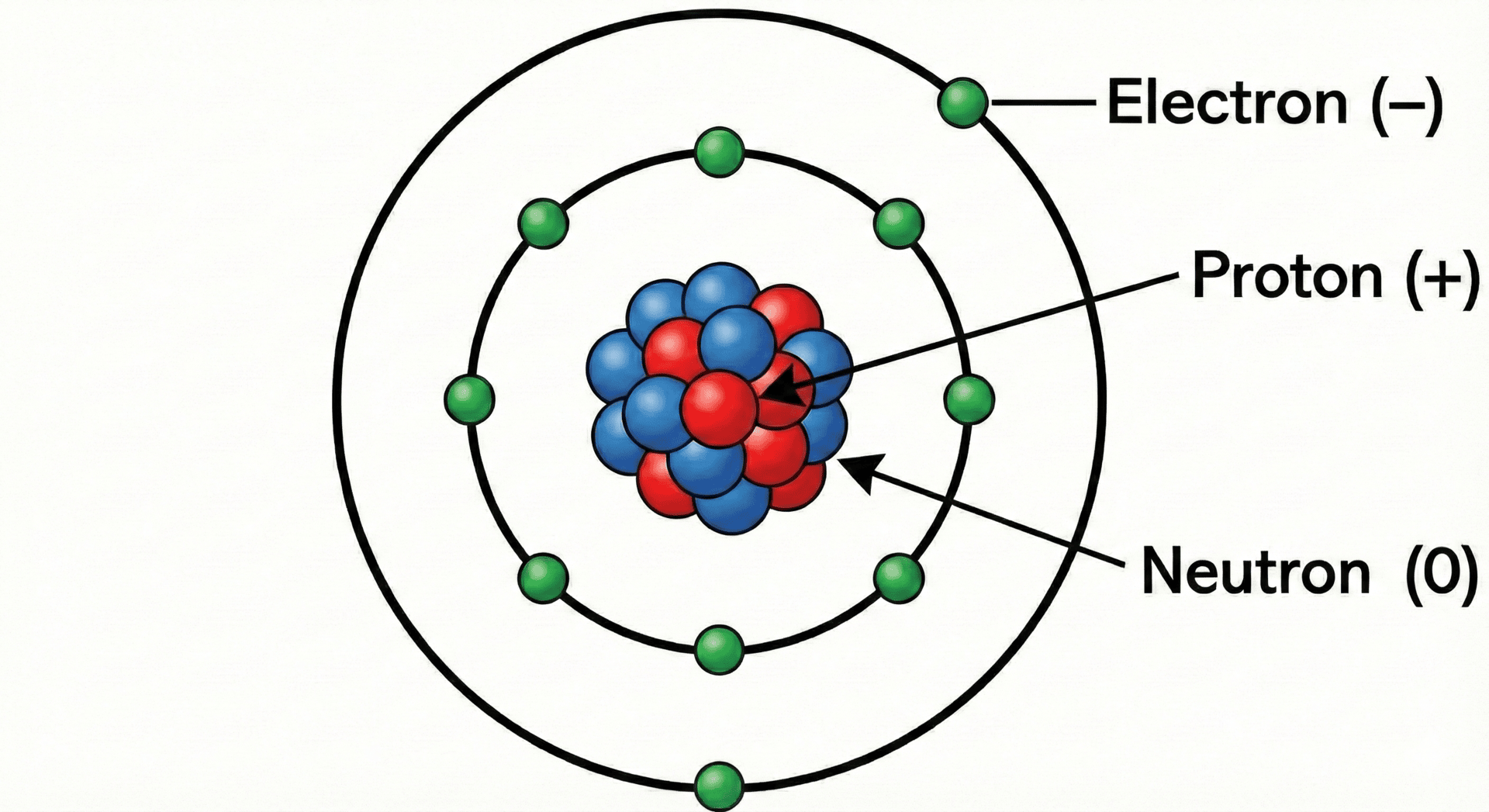
Subatomic Particles
The nucleus contains two types of particles: protons and neutrons. Collectively, these are known as nucleons. The electrons orbit the nucleus at a relatively large distance, meaning most of an atom is actually empty space.
Relative Mass and Charge
Because subatomic particles are so incredibly small, we use relative values to compare them rather than their actual masses in grams or charges in coulombs.
| Particle | Position | Relative Mass | Relative Charge |
|---|---|---|---|
| Proton | Nucleus | 1 | +1 |
| Neutron | Nucleus | 1 | 0 |
| Electron | Shells | 1/1840 (negligible) | -1 |

Atomic and Mass Numbers
Every element in the Periodic Table is defined by its unique atomic structure. We use two specific numbers to describe this:
1. Atomic Number (Z)
The atomic number is the number of protons in the nucleus of an atom.
- It defines the identity of the element (e.g., any atom with 6 protons is Carbon).
- In a neutral atom, the number of protons is equal to the number of electrons.
2. Mass Number (A)
The mass number is the total number of protons plus neutrons in the nucleus.
- Electrons are so light that their mass is ignored when calculating the mass number.
- You can find the number of neutrons in an atom using the formula: Number of Neutrons = Mass Number (A) - Atomic Number (Z)
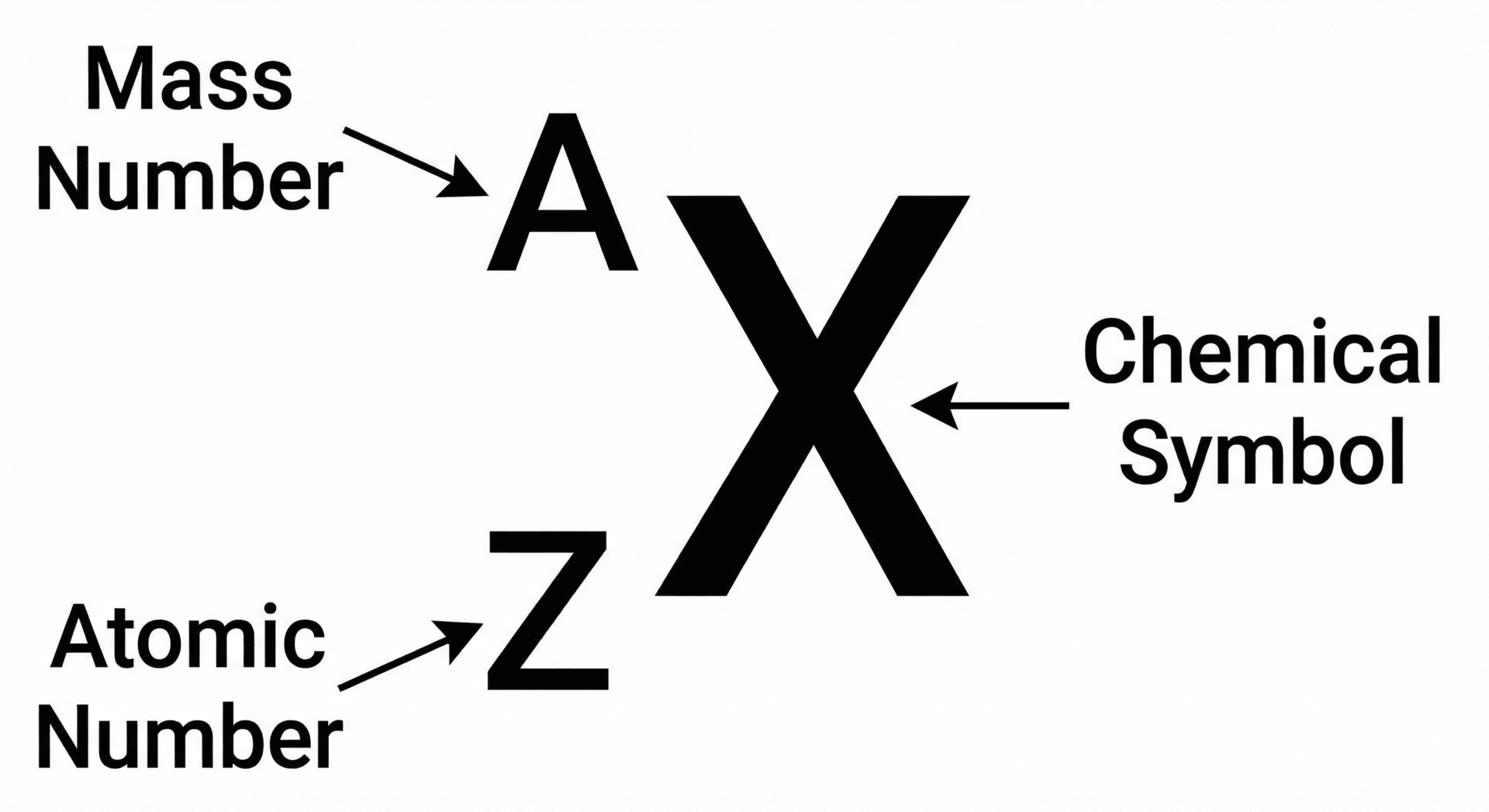
Practice Questions and Solutions
An atom of Potassium has a mass number of 39 and an atomic number of 19. State the number of protons, neutrons, and electrons present in a neutral atom of Potassium.
Protons: The atomic number is 19, so there are 19 protons.
Electrons: In a neutral atom, protons = electrons, so there are 19 electrons.
Neutrons: Mass Number - Atomic Number = 39 - 19 = 20 neutrons.
A Magnesium ion 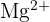 has an atomic number of 12 and a mass number of 24. Determine the number of electrons and neutrons in this ion.
has an atomic number of 12 and a mass number of 24. Determine the number of electrons and neutrons in this ion.
Neutrons: 24 - 12 = 12 neutrons.
Electrons: The atomic number is 12 (12 protons). A 2+ charge means the atom has lost 2 electrons. Therefore, 12 - 2 = 10 electrons.
An unknown neutral atom has 14 neutrons and 13 electrons. Identify the element and provide its mass number.
Protons: In a neutral atom, electrons = protons. Since there are 13 electrons, there are 13 protons.
Identity: Looking at the Periodic Table, the element with atomic number 13 is Aluminium (Al).
Mass Number: Protons + Neutrons = 13 + 14 = 27.
A neutral atom of Chlorine has a mass number of 35 and an atomic number of 17. State the number of protons, neutrons, and electrons present in this atom.
Protons: The atomic number is 17, so there are 17 protons.
Electrons: This is a neutral atom, so the number of electrons equals the number of protons: 17 electrons.
Neutrons: Subtract the atomic number from the mass number: 35 - 17 = 18 neutrons.
An unknown neutral atom has 4 neutrons and 3 electrons. Identify the element and provide its mass number.
Protons: In a neutral atom, the number of electrons is equal to the number of protons. Since there are 3 electrons, there are 3 protons.
Identity: Looking at the Periodic Table, the element with the atomic number 3 (3 protons) is Lithium (Li).Mass Number:
To find the mass number, add the protons and neutrons together: 3 protons + 4 neutrons = 7.
Summarise with AI:











