
Understanding Relative Mass
In chemistry, atoms are too small to weigh on a standard scale. Instead, we use the carbon-12 isotope as a standard, assigning it a mass of exactly 12. All other masses are measured relative to 1/12th the mass of one atom of carbon-12.
1. Relative Isotopic Mass
This is the mass of an atom of a specific isotope relative to 1/12th the mass of an atom of carbon-12.
- Because it is a ratio, it has no units.
- For most isotopes, this value is very close to the mass number (e.g., the relative isotopic mass of Chlorine-35 is approximately 35.0).
2. Relative Atomic Mass (Ar)
This is the weighted average mass of an atom of an element, taking into account its naturally occurring isotopes, relative to 1/12th the mass of an atom of carbon-12.
- To calculate Ar, you multiply the mass of each isotope by its percentage abundance and add them together.
Mass of Compounds
When atoms join together to form compounds, we sum their individual relative atomic masses.
3. Relative Molecular Mass (Mr)
This term is used for substances that exist as molecules (usually covalent compounds).
- It is the average mass of a molecule relative to 1/12th the mass of an atom of carbon-12.
- Example: For water
 , Mr = (2 x 1.0) + 16.0 = 18.0.
, Mr = (2 x 1.0) + 16.0 = 18.0.
4. Relative Formula Mass (Mr)
This term is used for ionic compounds (like NaCl) or giant covalent structures (like silicon dioxide) that don't exist as individual molecules.
- The calculation method is exactly the same as molecular mass: add up the Ar values of all the atoms shown in the empirical formula.
- Example: For Magnesium Chloride
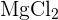 , Mr = 24.3 + (2 x 35.5) = 95.3.
, Mr = 24.3 + (2 x 35.5) = 95.3.
Practice Questions & Solutions
Antimony exists as two isotopes: 121Sb (abundance 57.3%) and 123Sb (abundance 42.7%). Calculate the relative atomic mass of Antimony to one decimal place.
Calculation: Ar =
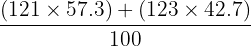
Step 2: Ar =
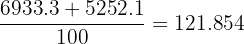
Final Answer: 121.9
Calculate the relative molecular mass (Mr) of ethanol, 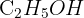 . (Ar: C = 12.0, H = 1.0, O = 16.0).
. (Ar: C = 12.0, H = 1.0, O = 16.0).
Calculation: (2 x 12.0) + (6 x 1.0) + (1 x 16.0)
Step 2: 24.0 + 6.0 + 16.0 = 46.0
Final Answer: 46.0
Determine the relative formula mass of 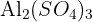 . (Ar: Al = 27.0, S = 32.1, O = 16.0).
. (Ar: Al = 27.0, S = 32.1, O = 16.0).
Calculation: (2 x 27.0) + 3[32.1 + (4 x 16.0)]
Step 2: 54.0 + 3(32.1 + 64.0) = 54.0 + 3(96.1)
Step 3: 54.0 + 288.3 = 342.3
Final Answer: 342.3
The relative atomic mass of an element with two isotopes is 10.8. One isotope is 10B (abundance 20.0%). What is the relative isotopic mass of the other isotope (abundance 80.0%)?
Step 1 Setup: 10.8 =
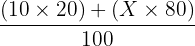
Step 2:
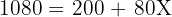
Step 3:
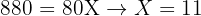
Final Answer: 11
Calculate the relative formula mass of hydrated copper(II) sulfate, 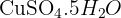 . (Ar: Cu = 63.5, S = 32.1, O = 16.0, H = 1.0).
. (Ar: Cu = 63.5, S = 32.1, O = 16.0, H = 1.0).
Note: The "dot" means you add the mass of 5 water molecules to the copper(II) sulfate.
Calculation: [63.5 + 32.1 + (4 x 16.0)] + 5[(2 x 1.0) + 16.0]
Step 2: [159.6] + 5[18.0] = 159.6 + 90.0
Final Answer: 249.6
Summarise with AI:











