Isotopes are atoms of the same element that have the same number of protons but a different number of neutrons. Because they have the same number of protons, they occupy the same position in the Periodic Table.
Key Characteristics:
- Proton Number (Z): Remains the same for all isotopes of a specific element.
- Neutron Number: Varies, which is what distinguishes one isotope from another.
- Mass Number (A): Changes because the total number of nucleons (protons + neutrons) is different.

Chemical vs. Physical Properties
One of the most important concepts in A-Level Chemistry is understanding why isotopes behave the way they do in different environments.
Chemical Properties
Isotopes of the same element have identical chemical properties.
- Chemical reactions involve the electrons, specifically those in the outer shell.
- Since isotopes of the same element have the same number of protons, they also have the same number of electrons in a neutral state.
- Therefore, they react in exactly the same way when forming bonds or undergoing chemical changes.
Physical Properties
Isotopes have different physical properties.
- Physical properties, such as density, melting point, and boiling point, often depend on the mass of the atom.
- Because isotopes have different numbers of neutrons, they have different masses.
- Heavier isotopes (those with more neutrons) typically have higher densities and slightly higher melting/boiling points.
Using the Mass Number Formula
Since the proton number (atomic number) is the "ID card" of an element, it never changes between isotopes. You can use this consistency to solve for unknown values using the standard formula:
Mass Number (A) = Protons (Z) + Neutrons
If you are given the mass number of a specific isotope and the number of neutrons, you can find the proton number to identify the element on the Periodic Table.
How to Write Isotopes
There are two primary ways to represent isotopes so that the specific mass is clear:
- Hyphen Notation: The name of the element followed by its mass number (e.g., Carbon-12 or Carbon-14).
- Nuclear Symbolism: Using the generic format where the mass number (A) is a superscript and the atomic number (Z) is a subscript to the left of the chemical symbol (X).
Practice Questions & Solutions
Define the term "isotope" and explain why Carbon-12 and Carbon-13 have the same chemical properties.
Definition: Isotopes are atoms of the same element with the same number of protons but different numbers of neutrons.
Reasoning: They have the same chemical properties because they have the same number and arrangement of electrons, which are responsible for chemical reactions.
An isotope of an unknown element has a mass number of 37 and contains 20 neutrons. Identify the element.
Calculate Protons: Protons = Mass Number - Neutrons = 37 - 20 = 17.
Identify: Element 17 is Chlorine (Cl).
Which isotope would you expect to have a higher density: Uranium-235 or Uranium-238? Explain your answer.
Answer: Uranium-238.
Reasoning: It has a higher mass number (more neutrons), and since density is mass per unit volume, the heavier isotope is denser.
Write the nuclear symbol for Chlorine-37. (Hint: Chlorine is element 17).
Answer:
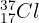
Calculate the number of neutrons in an atom of Magnesium-26 (Atomic number 12).
Calculation: Neutrons = 26 - 12 = 14.
Final Answer: 14 neutrons.
Summarise with AI:











