
Relative Atomic Mass and Isotopes
In chemistry, the Relative Atomic Mass (Ar) of an element is rarely a whole number because most elements exist as a mixture of different isotopes. A mass spectrometer is the primary tool used to determine the exact mass and relative abundance of these isotopes.
The Ar value we see on the Periodic Table is the weighted average of the masses of all naturally occurring isotopes relative to 1/12th the mass of an atom of Carbon-12.
How a Mass Spectrometer Works
A mass spectrometer works by converting atoms or molecules into ions so they can be manipulated by electric and magnetic fields. Here is the simplified process:
- Ionisation: The sample is bombarded with high-energy electrons from an "electron gun." These high-speed electrons "knock off" one or more electrons from the sample particles, creating positive ions.
- Acceleration: These positive ions are accelerated by an electric field so they all have the same kinetic energy.
- Deflection: The ions are then deflected by a magnetic field. Lighter ions and those with a higher charge are deflected more than heavier, lower-charged ions.
- Detection: The ions hit a detector, which records the mass-to-charge ratio (m/z) and the relative abundance of each ion.
Molecular Ions and the M+ Peak
When a whole molecule (M) loses an electron in the ionisation chamber, it forms a molecular ion (M+). The peak with the highest m/z value (excluding the M+1 peak) represents this molecular ion and tells us the relative molecular mass of the compound.
The M+1 Peak
You may notice a very small peak exactly one mass unit higher than the main molecular ion peak (M+). This is called the M+1 peak.
- It occurs because about 1.1% of all carbon atoms are the Carbon-13 isotope rather than Carbon-12.
- If a molecule contains at least one Carbon-13 atom, its total mass will be one unit higher, creating this tiny "ghost" peak.
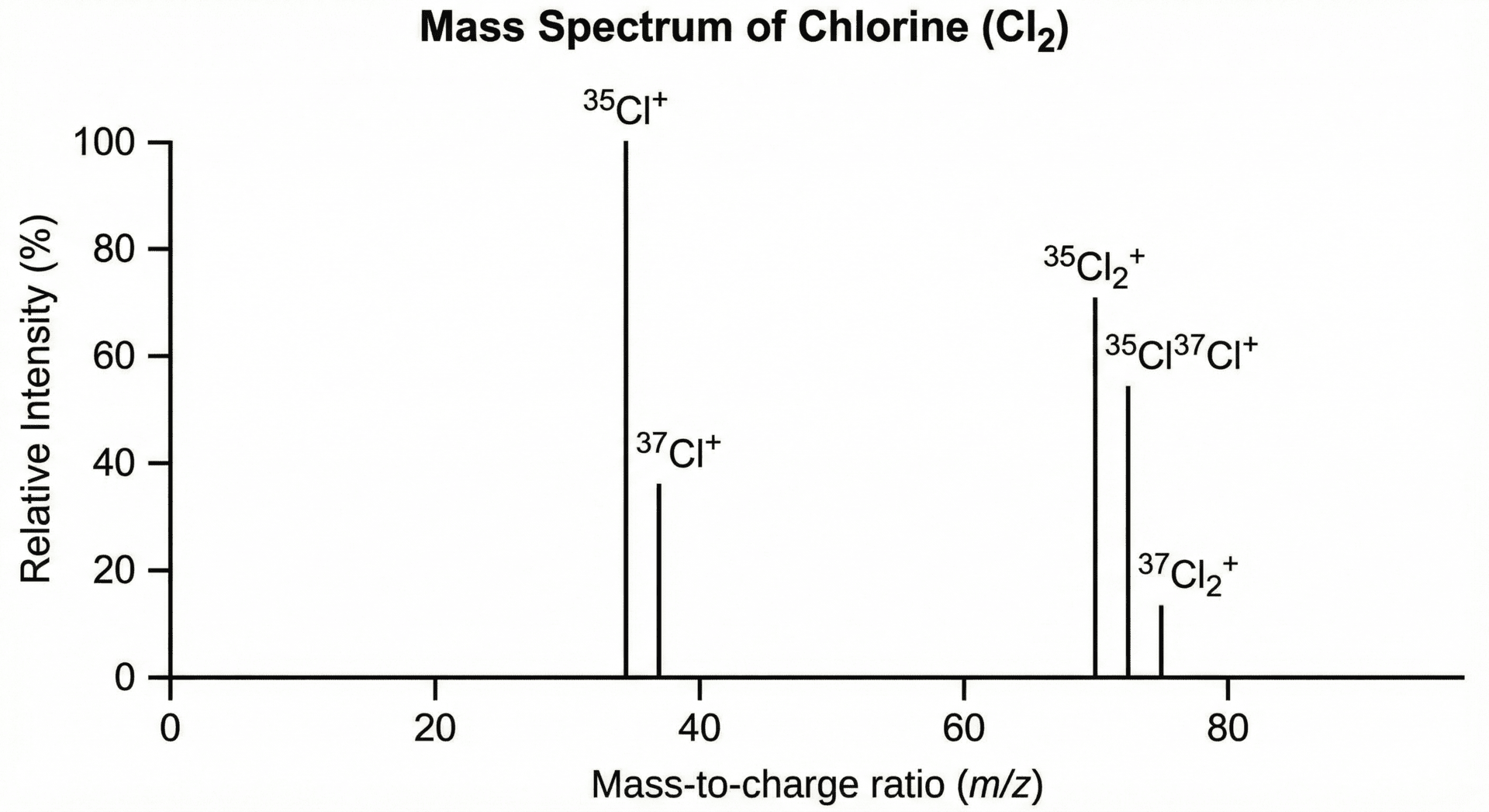
Case Study: The Mass Spectrum of Chlorine
Chlorine is a classic A-Level example because it exists as two main isotopes: Chlorine-35 (75% abundance) and Chlorine-37 (25% abundance).
Because chlorine is a diatomic molecule (formed of two atoms), the spectrum shows peaks for both individual atoms and the combined molecules:
- Atomic Peaks: You will see two peaks at m/z 35 and m/z 37 in a 3:1 height ratio.
- Molecular Peaks: When two chlorine atoms bond, they can form three different combinations:
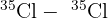 (Total mass = 70)
(Total mass = 70)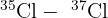 (Total mass = 72)
(Total mass = 72)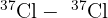 (Total mass = 74)
(Total mass = 74)
- The molecular peaks at 70, 72, and 74 appear in a characteristic 9:6:1 ratio.
Common Fragment Peaks
In the mass spectrometer, high-energy collisions often break molecules into smaller pieces called fragments. Identifying these fragments is like solving a puzzle to determine the molecule's structure. Some of the most common organic fragments are shown in the table below.
| m/z Value | Possible Fragment | Functional Group Clue |
|---|---|---|
| 15 | 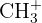 | Methyl group |
| 17 | 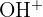 | Alcohol group |
| 28 | 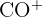 | Carbonyl group |
| 29 | 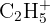 or or 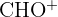 | Ethyl group or Aldehyde |
| 43 | 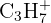 or or 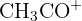 | Propyl group or Ketone |
| 45 | 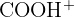 | Carboxylic acid |
Practice Questions & Solutions
In the mass spectrum of Propan-1-ol, what is the m/z value of the molecular ion peak?
Total mass =
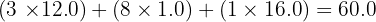
Final Answer: The molecular ion peak will be found at m/z 60.
Why is the peak at m/z 72 in the chlorine spectrum twice as high as the peak at m/z 74?
Combinations: The peak at m/z 72 represents the {35}Cl-{37}Cl molecule, while m/z 74 represents {37}Cl-{37}Cl.
Probability: Statistically, there are two ways to form the 72 molecule ({35}Cl then {37}Cl, or {37}Cl then {35}Cl), but only one way to form the 74 molecule ({37}Cl then {37}Cl).
Ratio: Because {35}Cl is three times more abundant than {37}Cl, the mathematical probability results in a 6:1 ratio between the 72 and 74 peaks.
Final Answer: The 72 peak is significantly higher (specifically 6 times higher) because the combination of a light and heavy isotope is statistically more likely than two heavy isotopes together.
A compound with the formula 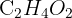 shows a strong fragment peak at m/z 45. Suggest the identity of this fragment and the functional group present.
shows a strong fragment peak at m/z 45. Suggest the identity of this fragment and the functional group present.
Formula: 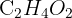 has a total mass of 60.
has a total mass of 60.
Identifying m/z 45: Subtracting 45 from the total mass (60 - 45) leaves a mass of 15, which corresponds to a lost methyl group 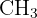
Fragment Identity: The fragment at m/z 45 is the 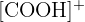 ion.
ion.
Functional Group: This fragment is characteristic of carboxylic acids.
Final Answer: The fragment is 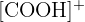 and the compound is likely ethanoic acid.
and the compound is likely ethanoic acid.
Summarise with AI:











