
Introduction
While ionic bonding involves the transfer of electrons and covalent bonding involves sharing them, metals employ a unique strategy known as Metallic Bonding. This bonding explains why metals are the backbone of modern infrastructure—from the copper wiring in your home to the steel beams in skyscrapers. By understanding how a giant lattice of positive ions exists within a sea of delocalised electrons, we can explain the remarkable conductivity, strength, and flexibility of metallic elements.
Theory
1. The Metallic Bonding Model
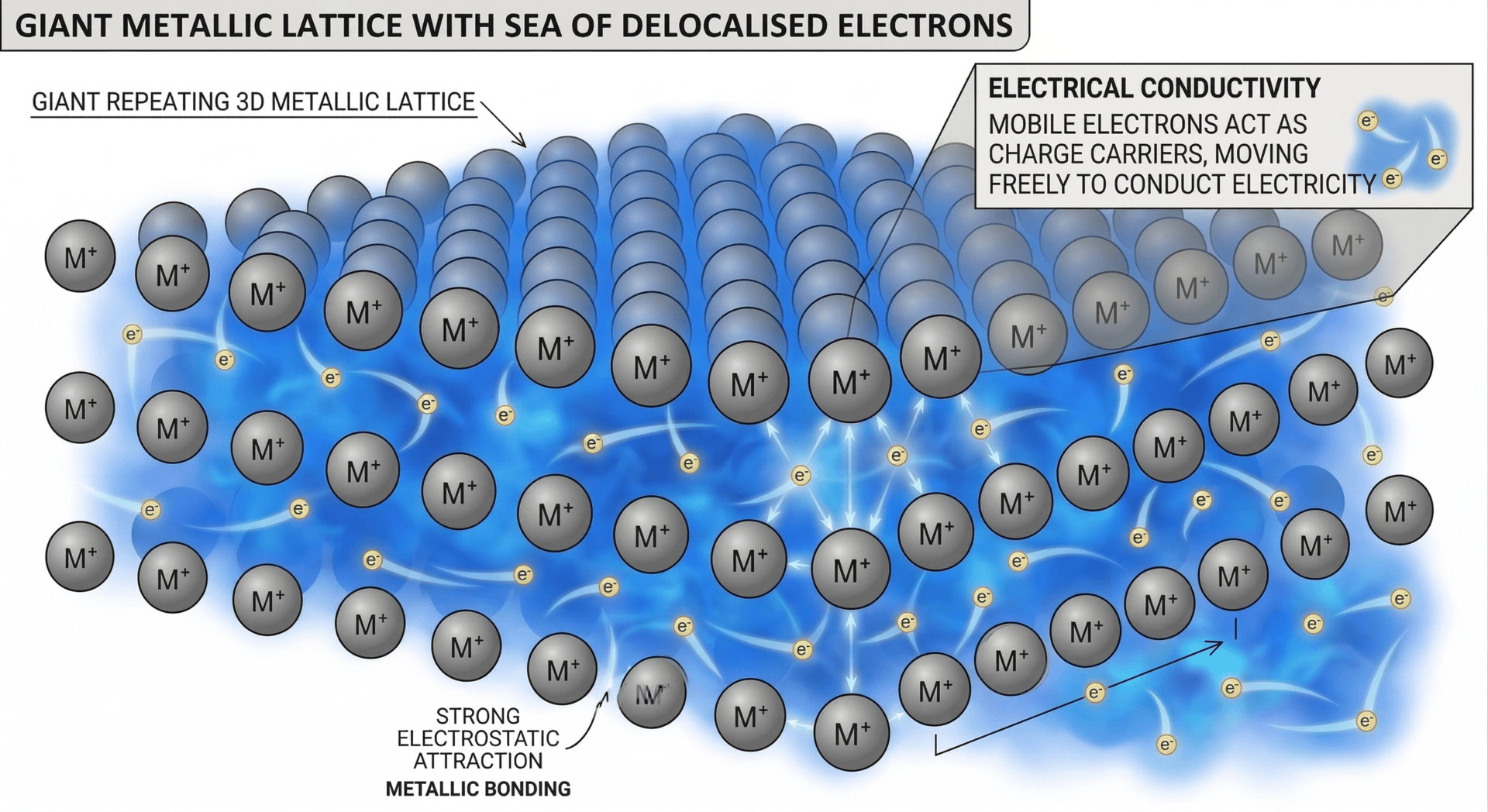
Metallic bonding is defined as the strong electrostatic attraction between a regular lattice of positive metal ions (cations) and a "sea" of delocalised electrons.
In a metallic structure, the atoms lose their outer-shell electrons to become positive ions. These electrons are no longer attached to any specific atom; instead, they are free to move throughout the entire 3D structure. We describe these electrons as being delocalised.
2. The Structure of the Giant Lattice
Metals form a giant metallic lattice. This is a highly ordered, repeating 3D arrangement of cations. The stability of this lattice is maintained by the pervasive attraction of the delocalised electrons, which act as a "glue" holding the positive ions in place.
3. Explaining Physical Properties
The "sea of electrons" model provides a clear explanation for the characteristic properties of metals:
| Property | Observation | Explanation |
|---|---|---|
| Electrical Conductivity | High | The delocalised electrons are mobile charge carriers that can move through the lattice when a potential difference is applied. |
| Thermal Conductivity | High | Mobile electrons can transfer kinetic energy (heat) rapidly throughout the structure. |
| Malleability & Ductility | High | The layers of positive ions can slide over each other without breaking the bond because the "sea" of electrons shifts to maintain the attraction. |
| Melting & Boiling Points | Generally High | Strong electrostatic attractions between the cations and the delocalised electrons require significant energy to overcome. |
4. Factors Affecting Bond Strength
Not all metallic bonds are of equal strength. The strength of the metallic bond depends on:
- The Number of Delocalised Electrons: The more electrons an atom contributes to the sea (e.g., Al contributes 3, while Na contributes 1), the stronger the electrostatic attraction.
- The Charge of the Cation: A higher positive charge (e.g., Mg(2+) vs Na(+)) creates a stronger pull on the delocalised electrons.
- The Size of the Cation: Smaller ions have a higher charge density, allowing the delocalised electrons to get closer to the nucleus, increasing the attraction.
Worked Example
Question: Explain why Aluminium has a significantly higher melting point than Sodium.
Step 1: Compare the number of delocalised electrons - Sodium is in Group 1 and contributes one electron per atom to the delocalised sea. Aluminium is in Group 13 and contributes three electrons per atom.
Step 2: Compare the ionic charge and size - The Sodium ion is Na(+), while the Aluminium ion is Al(3+). Additionally, Al(3+) is smaller than Na(+) because it has a higher nuclear charge pulling on the same number of electron shells.
Step 3: Relate to bond strength - Aluminium has a higher density of delocalised electrons and a cation with a higher charge and smaller radius. This results in a much stronger electrostatic attraction between the ions and the electrons.
Step 4: Conclusion - Because the metallic bonding in Aluminium is much stronger than in Sodium, significantly more thermal energy is required to break the lattice. Therefore, Aluminium has a higher melting point.
Practice Questions & Solutions
Define metallic bonding in terms of electrostatic forces.
Metallic bonding is the strong electrostatic attraction between positive metal ions and delocalised electrons.
Explain why metals are described as being malleable.
The positive ions are arranged in regular layers. When a force is applied, these layers can slide over one another. The delocalised electrons move with the layers, maintaining the metallic bond throughout the movement.
Predict the trend in melting points for the Group 1 metals (alkali metals) as you move down the group.
The melting point decreases down the group. As you move down, the ionic radius of the cations increases. The electrostatic attraction between the nucleus and the delocalised electrons becomes weaker as the distance increases.
State the formula of the ion formed by Magnesium in a metallic lattice and the number of electrons it contributes to the sea.
The ion formed is Mg(2+). It contributes 2 electrons per atom to the delocalised sea.
Why can metals conduct electricity in the solid state while ionic compounds cannot?
In a metal, delocalised electrons are mobile charge carriers even in the solid state. In a solid ionic compound, the ions are held in fixed positions and cannot move.
Related Articles:
Continue your A-Level Chemistry Revision with the following articles:
Summarise with AI:











