At A-Level, we move beyond the idea of electrons "orbiting" the nucleus like planets. Instead, we describe the position of an electron in terms of probability. An atomic orbital is a specific 3D region of space around the nucleus where there is a high probability (usually around 95%) of finding an electron.

What Are Atomic Orbitals?
Key Orbital Principles
- Energy Levels: Orbitals exist at specific energy levels (shells). Electrons can only exist within these levels, never in the gaps between them.
- Maximum Capacity: Every single atomic orbital can hold a maximum of two electrons.
- Subshell Structure: The number of orbitals differs depending on the subshell type:
| Subshell | Number of Orbitals | Total Electron Capacity |
|---|---|---|
| s | 1 | 2 |
| p | 3 | 6 |
| d | 5 | 10 |
| f | 7 | 14 |
Orbital Shapes
You are required to understand and visualize the 3D shapes of s and p orbitals. (Note: You do not need to know the shapes of d orbitals for this specification).
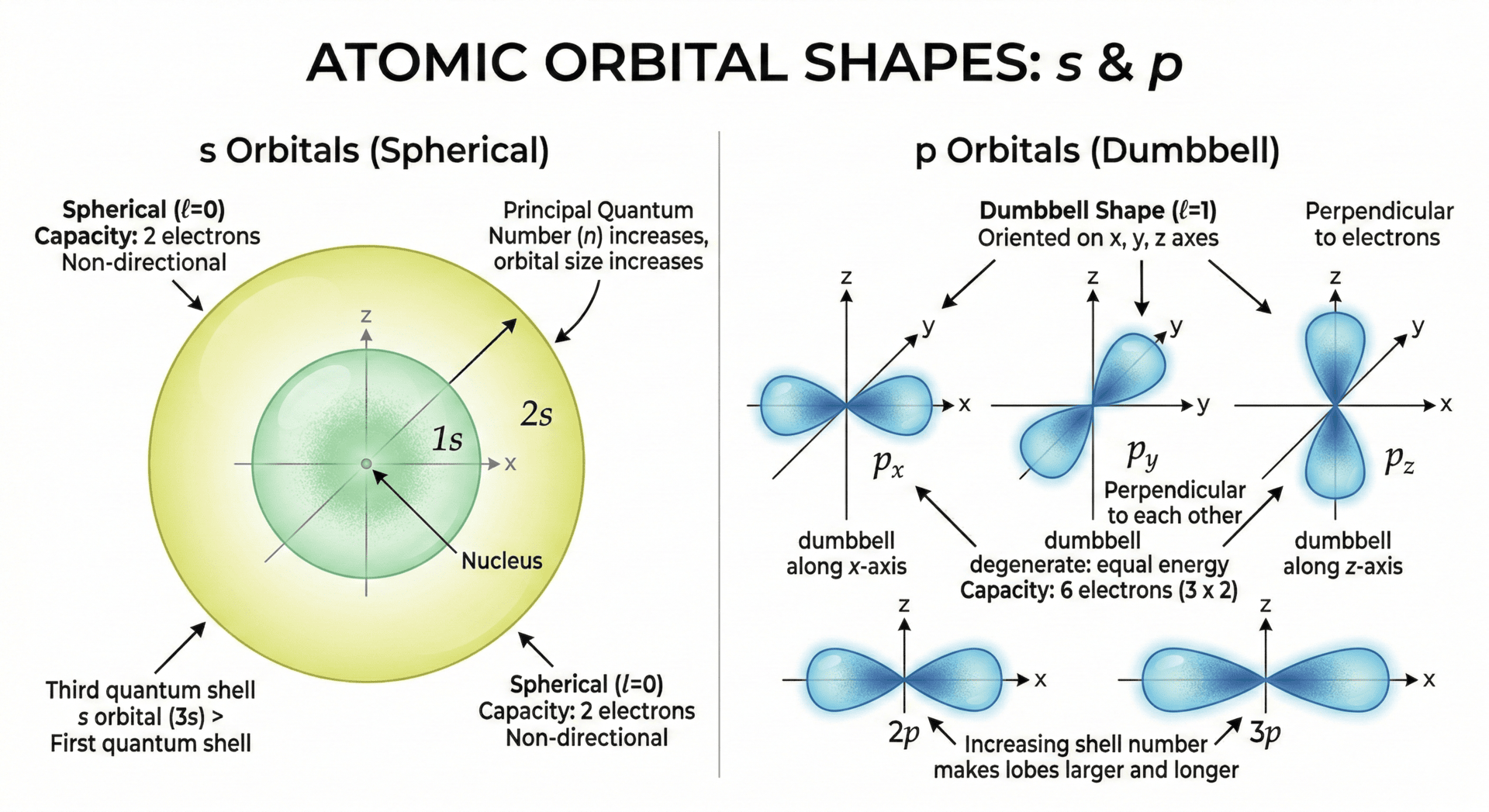
s Orbitals: The Sphere
The s orbital is perfectly spherical in shape. The nucleus is located at the very center of this sphere.
- Size vs. Shell Number: As the principal quantum number (n) increases, the s orbital becomes larger. For example, a 3s orbital is significantly larger and further from the nucleus than a 1s orbital.
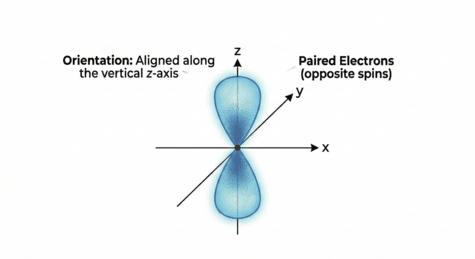
p Orbitals: The Dumbbell
Starting from the second shell (n = 2), every energy level contains three p orbitals.
- Shape: These have a "dumbbell" or "lobed" shape.
- Orientation: The three p orbitals are oriented at right angles to each other along the x, y, and z axes. They are labeled as
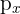 ,
, 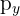 , and
, and 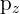 .
. - Size vs. Shell Number: Similar to s orbitals, the lobes of p orbitals become longer and larger as you move to higher shells.
Filling Orbitals
To understand how electrons inhabit these orbitals, we must look at how they behave as "spinning charges."
Electron Spin
Electrons rotate around their own axis in either a clockwise or anticlockwise direction. This spin creates a tiny magnetic field.
- Spin-Pair Repulsion: Electrons with the same spin repel each other.
- Opposite Spins: To occupy the same orbital, two electrons must have opposite spins (represented by an up arrow and a down arrow). This is known as the Pauli Exclusion Principle.
Hund’s Rule
When electrons are filling a subshell with multiple orbitals (like the p subshell), they follow a specific order to minimise repulsion:
- Electrons will occupy separate orbitals singly first, keeping their spins in the same direction.
- Only once every orbital in that subshell has one electron will they begin to pair up.
For example, if a p subshell has three electrons, one will go into each of the 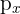 ,
, 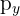 , and
, and 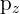 orbitals rather than two pairing up in the first one.
orbitals rather than two pairing up in the first one.
Exam Focus: Common Pitfalls
- The Energy Gap: Why do electrons pair up at all if they repel each other? Because the energy required to jump to a completely different, higher-energy empty orbital is much greater than the "cost" of the repulsion within a lower-energy orbital.
- Labelling: In diagrams, always ensure you label your axes (x, y, z) when drawing p orbitals to show they are perpendicular.
- Arrows: Always draw your arrows in opposite directions within a single box to represent the Pauli Exclusion Principle.
Practice Questions & Solutions
Define an "atomic orbital."
A region of space around the nucleus where there is a high probability of finding an electron.
State the maximum number of electrons that can be held in a 3d subshell.
A d subshell has 5 orbitals, and each orbital holds 2 electrons. Therefore, the total is 10 electrons.
Explain why the two electrons in a 2s orbital must have opposite spins.
To minimize the electrostatic repulsion between them (spin-pair repulsion) and to comply with the Pauli Exclusion Principle.
Draw the shape of a 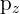 orbital.
orbital.
Using Hund's Rule, describe the arrangement of 4 electrons in a 3p subshell.
Three electrons will occupy the 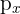 ,
, 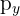 , and
, and 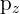 orbitals singly with parallel spins. The fourth electron will then pair up in the
orbitals singly with parallel spins. The fourth electron will then pair up in the 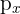 orbital with an opposite spin.
orbital with an opposite spin.
Related Articles:
Continue your A-Level Chemistry Revision with the following articles:
- Physical Chemistry: Ionisation Energy
- Physical Chemistry: Electronic Configuration
- Physical Chemistry: Quantum Shells
Summarise with AI:











