Ionisation energy is a measure of the energy required to remove electrons from atoms or ions. Because electrons are negatively charged and are attracted to the positively charged nucleus, energy must be supplied to overcome this electrostatic attraction.
First Ionisation Energy
The first ionisation energy (IE) is the energy required to remove one mole of electrons from one mole of gaseous atoms to form one mole of gaseous 1+ ions.
The general equation is:
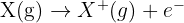
Successive Ionisation Energies
You can continue to remove electrons from the resulting ions. The second ionisation energy is the energy required to remove one mole of electrons from one mole of gaseous 1+ ions to form one mole of gaseous 2+ ions.
The second ionisation energy equation is:
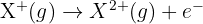
Exam Tip: Successive ionisation energies always increase because it is significantly harder to remove a negative electron from an increasingly positive ion.

Factors Affecting Ionisation Energy
The strength of the attraction between the nucleus and the outer electrons depends on three main factors:
- Nuclear Charge: The more protons in the nucleus, the greater the positive charge. This creates a stronger attraction for outer electrons.
- Atomic Radius: As the distance between the nucleus and the outer electrons increases, the electrostatic attraction weakens.
- Shielding: Inner shells of electrons "shield" the outer electrons from the full pull of the nucleus. More inner shells mean more shielding and a lower ionisation energy.
Trends Across a Period
Across any period, there is a clear pattern in first ionisation energy. While the general trend is an increase, there are specific "dips" that provide evidence for subshells and orbitals.
The General Trend: An Increase
As you move from left to right across a period (e.g., Sodium to Argon), the first ionisation energy generally increases. This is because:
- Nuclear Charge increases (more protons).
- Shielding remains constant (electrons are added to the same shell).
- Atomic Radius decreases (the nucleus pulls the shell closer).
The Exceptions (The "Dips")
Looking at row 3 of the periodic table, there are two characteristic drops at Group 3 and Group 6.
Dip 1: Group 2 to Group 3 (e.g., Mg to Al)
There is a slight drop because the outer electron in Aluminium is in a 3p subshell, which is at a higher energy level than the 3s subshell in Magnesium. The 3p electron is also slightly shielded by the 3s electrons, making it easier to remove.
Dip 2: Group 5 to Group 6 (e.g., P to S)
This drop is due to spin-pair repulsion. In Phosphorus (Group 5), the 3p orbitals are all occupied by single electrons. In Sulfur (Group 6), two electrons must share one 3p orbital. These two negative electrons repel each other, making it easier to remove one of them.
Successive Ionisation Energy Graphs
Successive ionisation energy data provides direct evidence for the existence of electron shells.
- Steady Increase: A gradual increase as you remove electrons from the same shell.
- Big Jumps: A sudden, large increase indicates that an electron is being removed from a shell closer to the nucleus (a lower principal quantum number).
A great visualisation for successive ionisaiton energies is that of Sodium, shown below:
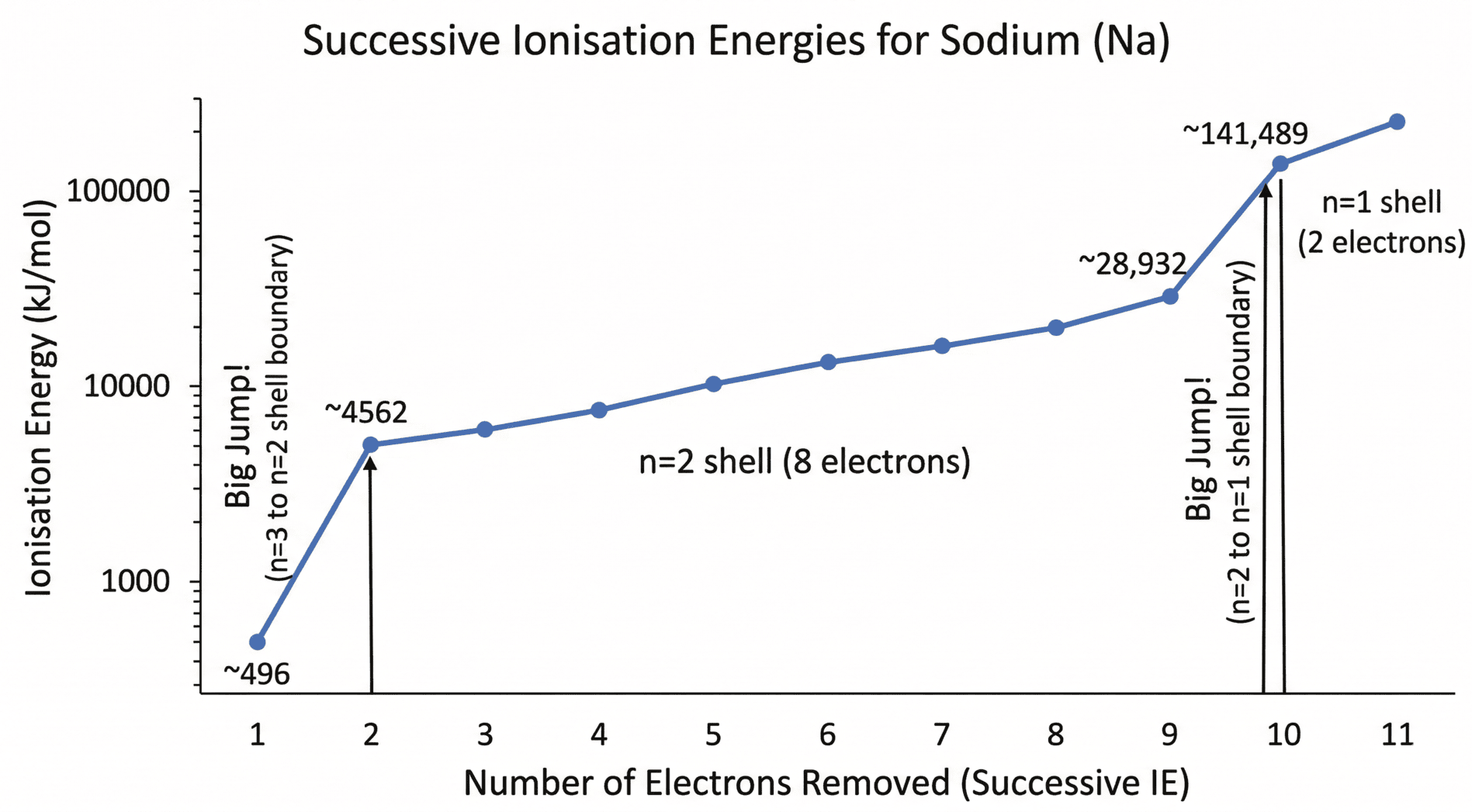
- Low 1st IE: Representing the removal of the outer 3s¹ electron.
- Huge Jump: From the 1st to the 2nd IE, marking the boundary between the n=3 and n=2 shells.
- Gradual Increase: Among the 8 electrons in the inner n=2 shell.
- Second Huge Jump: Between the 9th and 10th IE, marking the boundary between the n=2 and n=1 shells.
Practice Questions & Solutions
Define the term "First Ionisation Energy" and write an equation, including state symbols, for the first ionisation energy of Magnesium.
Definition: The energy required to remove one mole of electrons from one mole of gaseous atoms to form one mole of gaseous 1+ ions.
Equation:
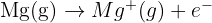
Explain why there is a general increase in first ionisation energy across Period 3 from Sodium to Argon.
Across Period 3, the number of protons increases, leading to a higher nuclear charge. The electrons are added to the same principal energy level, meaning shielding remains constant. This results in a smaller atomic radius and a stronger electrostatic attraction between the nucleus and the outer electrons.
The successive ionisation energies (in kJ/mol) for an unknown Period 3 element are: 578, 1817, 2745, 11577, 14842, 18379. Identify the element and explain your reasoning.
Identity: Aluminium (Al).
Reasoning: There is a massive jump between the 3rd and 4th ionisation energies (from 2745 to 11577 kJ/mol). This indicates that the fourth electron is being removed from a shell closer to the nucleus (n = 2). Therefore, the element has 3 electrons in its outer shell (Group 3). In Period 3, this is Aluminium.
Explain why the first ionisation energy of Boron (Z = 5) is lower than that of Beryllium (Z = 4), even though Boron has a higher nuclear charge.
Beryllium has the configuration:
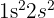
while Boron is:
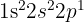 .
.
The outer electron in Boron is in the 2p subshell, which is at a higher energy level than the 2s subshell of Beryllium. The 2p electron also experiences more shielding from the 2s electrons, making it easier to remove.
Write the equation for the second ionisation energy of Sodium and explain why this value (4562 kJ/mol) is much higher than Sodium's first ionisation energy (496 kJ/mol).
Equation:
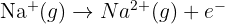
Explanation: The first electron is removed from the 3s subshell (n = 3). The second electron must be removed from the 2p subshell (n = 2), which is much closer to the nucleus and experiences significantly less shielding. The electrostatic attraction is much stronger, requiring far more energy.
Related Articles:
Continue your A-Level Chemistry Revision with the following articles:
- Physical Chemistry: Atomic Orbitals
- Physical Chemistry: Electronic Configuration
- Physical Chemistry - Quantum Shells
Summarise with AI:











