Ionic bonding is often the first type of chemical bonding students encounter, typically described as a simple "transfer of electrons." However, at A-Level, we move beyond this simplified model to explore the Ionic Trends that govern the strength and behaviour of these compounds.
Why does Magnesium Oxide have a significantly higher melting point than Sodium Chloride? Why do some ionic compounds exhibit "covalent character"? By examining trends in ionic radii, lattice energy, and polarisability, we can predict the physical properties of materials and understand the limits of the purely ionic model. This guide provides an in-depth look at these trends, aligned with AQA, OCR, and Edexcel specifications.

Theory
1. The Nature of the Ionic Bond
An ionic bond is the strong electrostatic attraction between oppositely charged ions (cations and anions). In a solid state, these ions arrange themselves into a giant ionic lattice, a regular repeating 3D structure that maximises the attraction between opposite charges while minimising the repulsion between like charges.
The strength of this attraction is not uniform across all compounds. It depends heavily on two fundamental periodic trends: ionic charge and ionic radius.
2. Trends in Ionic Radii
The ionic radius is the measure of an ion's size in a crystal lattice. Understanding how this size changes is crucial for predicting the strength of the ionic bond.
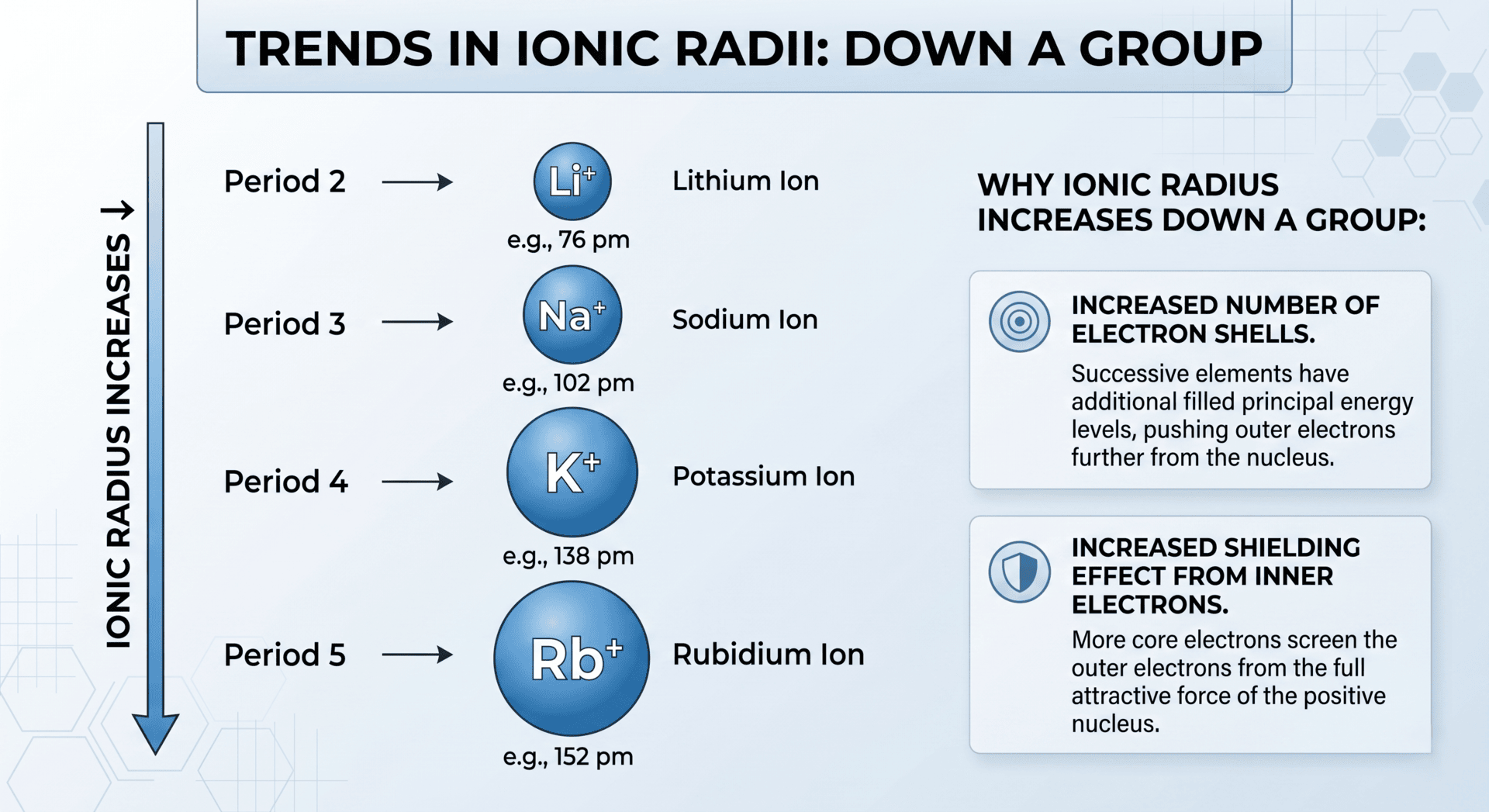
Down a Group
As you move down a group in the Periodic Table, the ionic radius increases.
- Each successive element has an additional occupied electron shell.
- The increased shielding from inner shells outweighs the increase in nuclear charge, resulting in a larger distance between the nucleus and the valence electrons.
Across an Isoelectronic Series
An isoelectronic series consists of ions that have the same number of electrons eg., the following all have 10 electrons:
- Across this series, the nuclear charge (number of protons) increases.
- Because the number of electrons remains constant, the increasing "pull" from the nucleus draws the electron clouds closer.
- Therefore, in an isoelectronic series, the ionic radius decreases as the atomic number increases.
3. Lattice Energy
Lattice energy is a quantitative measure of the strength of an ionic bond. It is defined as the enthalpy change when one mole of an ionic solid is formed from its constituent gaseous ions under standard conditions:
and can be represented as:
Lattice energy is always exothermic (negative value) because bond formation releases energy. A more negative (larger magnitude) lattice energy indicates a stronger ionic bond and a more stable lattice.
Factors Affecting Lattice Energy
The magnitude of lattice energy is governed by two main factors:
- Ionic Charge: The higher the charge on the ions, the stronger the electrostatic attraction. For example, Mg(2+) has a much stronger attraction to O(2-) than Na(+) has to Cl(-).
- Ionic Radius: Smaller ions can get closer to one another in the lattice. The distance between the centres of the ions is smaller, which increases the strength of the electrostatic attraction.
We can summarise the relationship using the following proportionality:
Where q represents the charges and r represents the sum of the ionic radii.
| Compound | Ionic Radii Sum (pm) | Lattice Energy (kJ/mol) | Melting Point (°C) |
|---|---|---|---|
| LiF | 201 | -1030 | 845 |
| LiCl | 257 | -853 | 605 |
| LiBr | 275 | -807 | 550 |
| LiI | 302 | -757 | 449 |
4. Polarisation and Covalent Character
The "purely ionic model" assumes that ions are perfect, undeformable spheres. In reality, many ionic bonds have a degree of covalent character. This occurs through a process called polarisation.
- Polarising Power: The ability of a cation to distort the electron cloud of an anion. Cations with a high charge density (small radius and high charge) have the greatest polarising power.
- Polarisability: The ease with which an anion's electron cloud can be distorted. Large anions with a high negative charge are the most polarisable because their outer electrons are further from the nucleus and less tightly held.
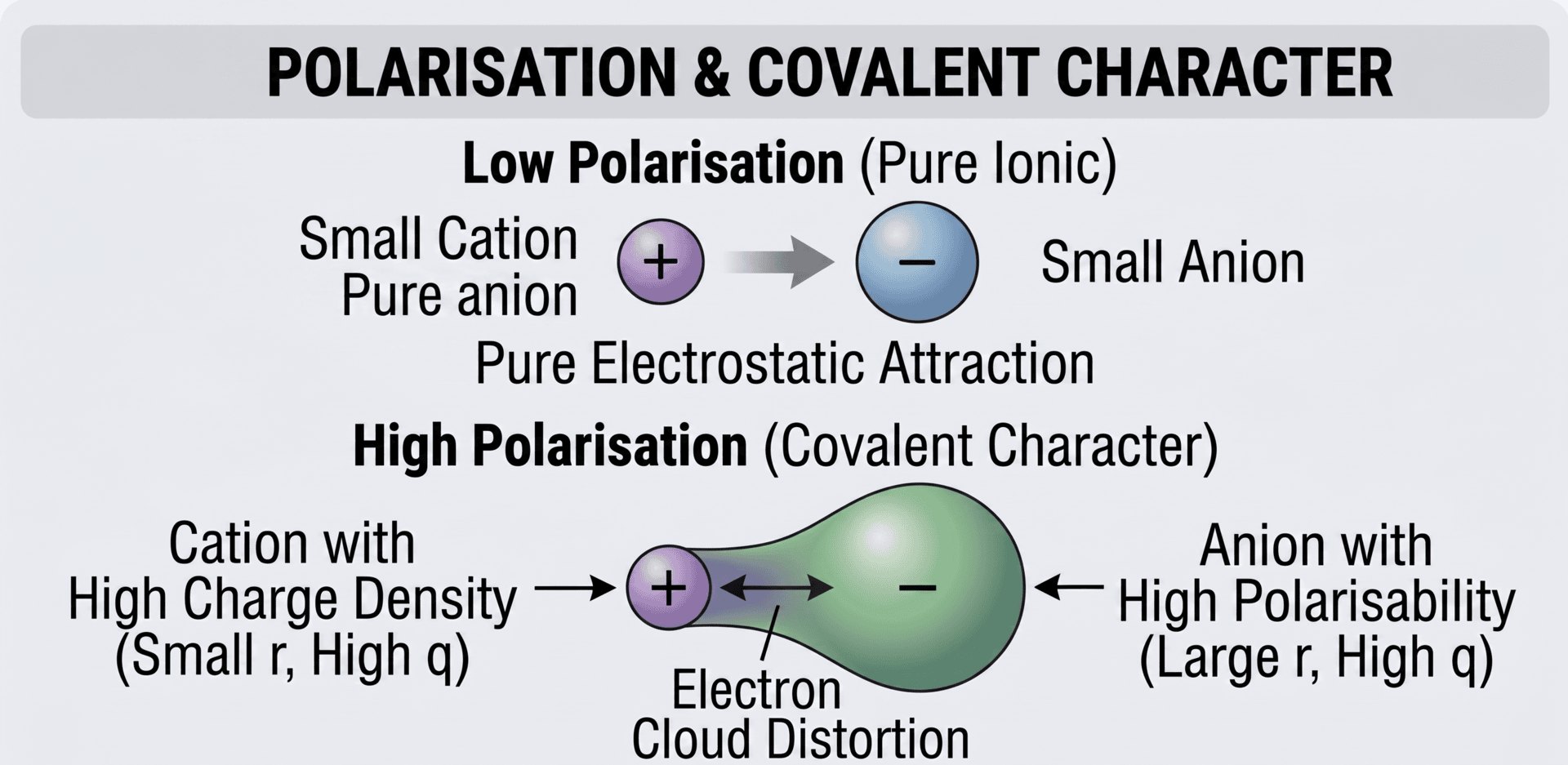
Fajan’s Rules
Fajan’s Rules help us predict when an ionic bond will show significant covalent character:
- The cation has a high charge and a small radius (high charge density).
- The anion has a high charge and a large radius.
When polarisation is extreme, the electron density is shared between the two nuclei rather than being completely transferred, leading to covalent character. This often results in melting points being lower than predicted by the purely ionic model.
Worked Example
Question: Compare the lattice energies of Magnesium Oxide (MgO) and Sodium Chloride (NaCl) and explain which will have the higher melting point.
Step 1 - Compare the ionic charges: In MgO, the ions are Mg(2+) and O2(-). In NaCl, the ions are Na(+) and Cl(-). The product of the charges in MgO (2 x 2 = 4) is four times greater than in NaCl (1 x 1 = 1).
Step 2 - Compare the ionic radii: Mg(2+) is smaller than Na(+) (it has more protons pulling on the same number of electron shells). O(2-) is smaller than Cl(-) (it has fewer electron shells). Therefore, the ions in MgO can get much closer together than the ions in NaCl.
Step 3 - Relate to Lattice Energy: Because MgO has higher charges and smaller ionic radii, the electrostatic attraction is significantly stronger. This results in a much more exothermic (larger negative) lattice energy for MgO.
Step 4 - Conclusion: MgO has a much higher lattice energy, meaning more energy is required to overcome the electrostatic forces of attraction to melt the solid. Therefore, MgO has a significantly higher melting point (approx. 2852°C) compared to NaCl (approx. 801°C).
Practice Questions & Solutions
Define the term 'isoelectronic' and arrange the following ions in order of decreasing ionic radius:
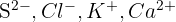
Isoelectronic ions have the same number of electrons. In this series, all have 18 electrons. As nuclear charge increases, the radius decreases. Order of decreasing radius:
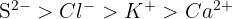
Which compound would you expect to have the most exothermic lattice energy: LiF, LiCl, or KF? Explain your answer.
LiF would have the most exothermic lattice energy. Lithium and Fluoride are the smallest ions in the set. Smaller ionic radii allow ions to get closer, increasing attraction.
Using Fajan's Rules, explain why Aluminium Iodide exhibits significant covalent character.
Aluminium Iodide contains a highly polarising cation and a highly polarisable anion. Al(3+) has a high charge and small radius (high charge density). I(-) is a very large anion with a distorable electron cloud.
Calculate the difference in the sum of ionic radii for NaCl and KCl given the following data:
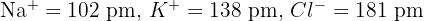
Calculate the sum for each:
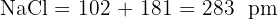
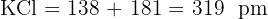
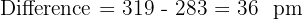
Explain why the experimental lattice energy for Silver Iodide (AgI) is often different from the theoretical value calculated using the purely ionic model.
The purely ionic model assumes ions are perfectly spherical. AgI has significant covalent character due to polarisation. The Ag(+) ion is highly polarising and the I(-) ion is highly polarisable. Extra bonding strength from electron sharing makes the experimental value more exothermic.
Related Articles:
Continue your A-Level Chemistry Revision with the following articles:
Summarise with AI:











