
Introduction
In the world of Chemistry, bonding is the force that holds atoms together to form the substances we see every day. Ionic bonding is a fundamental type of chemical attraction that occurs between metals and non-metals. At A-Level, we move beyond the simple idea of "giving and taking" electrons to explore the electrostatic forces and giant 3D structures that give ionic compounds their unique physical properties, such as high melting points and electrical conductivity in specific states.
Theory
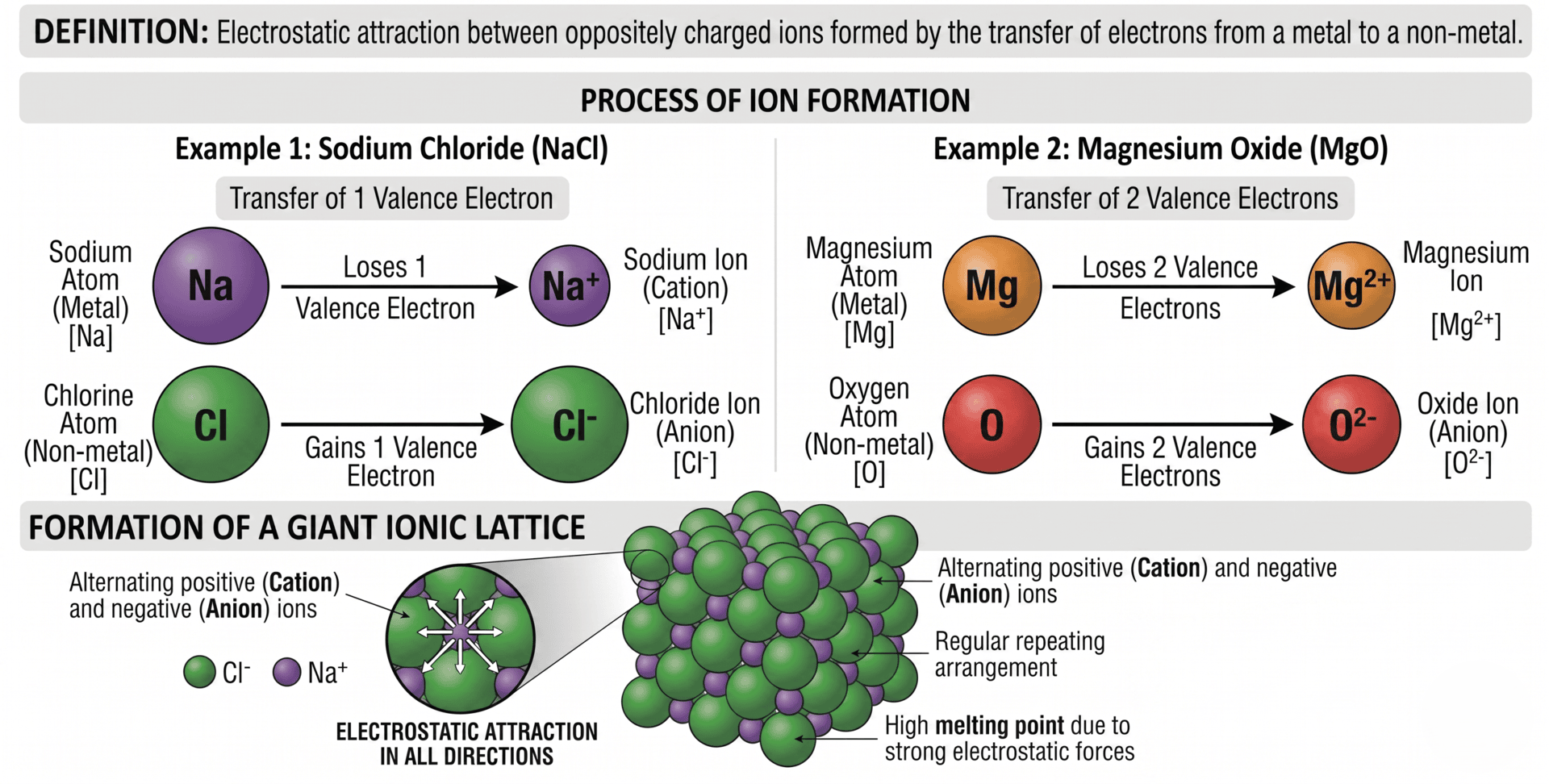
1. The Formation of Ions
Ionic bonding occurs when electrons are transferred from a metal atom to a non-metal atom. This process is driven by the atoms' "desire" to achieve a stable noble gas electron configuration (a full outer shell).
- Cations: Metal atoms lose electrons to form positively charged ions. For example, a Sodium atom (group 1) loses one electron to become Na(+).
- Anions: Non-metal atoms gain electrons to form negatively charged ions. For example, a Chlorine atom (group 17) gains one electron to become Cl(-).
The resulting ions are often isoelectronic with the nearest noble gas, meaning they have the same number of electrons and the same electronic configuration.
2. The Nature of the Ionic Bond
The ionic bond itself is defined as the strong electrostatic attraction between oppositely charged ions. This attraction acts in all directions (it is non-directional), meaning each positive ion attracts all surrounding negative ions and vice versa.
3. The Giant Ionic Lattice
Ionic compounds do not exist as isolated molecules. Instead, they form a giant ionic lattice. This is a regular, repeating three-dimensional arrangement where each ion is surrounded by ions of the opposite charge to maximise attraction and minimise repulsion.
- Coordination Number: This refers to the number of ions of opposite charge surrounding a central ion. In Sodium Chloride (NaCl), the coordination number is 6:6, meaning each Na(+) is surrounded by 6 Cl(-) ions, and each Cl(-) is surrounded by Na(+) ions.
4. Physical Properties of Ionic Compounds
The structure and bonding of ionic compounds directly dictate their physical properties:
| Property | Observation | Explanation |
|---|---|---|
| Melting and Boiling Points | High | Large amounts of energy are required to break the strong electrostatic attractions throughout the giant lattice. |
| Electrical Conductivity (Solid) | None | Ions are held in fixed positions within the lattice and cannot move to carry a charge. |
| Electrical Conductivity (Molten/Aqueous) | Good | The lattice breaks down and the ions are free to move (mobile ions) to act as charge carriers. |
| Brittleness | High | Applying a force shifts layers of ions. Like-charges then align and repel each other causing the crystal to shatter. |
| Solubility | Often Soluble in Water | Water molecules are polar and can attract the ions out of the lattice into the solution. |
5. Evidence for the Existence of Ions
We can prove the existence of ions through electron density maps. X-ray crystallography shows that in an ionic compound, the electron density drops to zero between the nuclei of the two ions. This confirms that the electrons are not shared (as in covalent bonding) but are entirely localised around the individual nuclei.
Worked Example
Task: Use dot-and-cross diagrams to show the formation of Magnesium Chloride and explain the resulting charges.
Step 1: Identify the initial electronic configurations
- Magnesium (Group 2): 2, 8, 2
- Chlorine (Group 17): 2, 8, 7
Step 2: Determine electron transfer: Magnesium needs to lose 2 electrons to achieve a full outer shell. Each Chlorine atom only needs 1 electron. Therefore, one Magnesium atom reacts with two Chlorine atoms.
Step 3: Write the ion configurations
- Magnesium becomes Mg(2+) (configuration: 2, 8).
- Each Chlorine becomes Cl(-) (configuration: 2, 8, 8).
Step 4: Summary of attraction The one Mg(2+) cation and two Cl(-) anions are held together by strong electrostatic attractions in a giant lattice with the empirical formula:
Practice Questions & Solutions
Define the term 'ionic bond'.
An ionic bond is the strong electrostatic attraction between oppositely charged ions.
Write the electronic configuration of the Al(3+) ion and state which noble gas it is isoelectronic with.
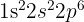
It is isoelectronic with Neon.
Explain why Magnesium Oxide, (MgO), has a much higher melting point than Sodium Chloride (NaCl).
The ions in MgO are Mg(2+) and O(2-), whereas in NaCl they are Na(+) and Cl(-). The higher charges on the ions in MgO result in much stronger electrostatic attractions. More energy is required to overcome these stronger forces in the MgO lattice.
State the conditions required for an ionic compound to conduct electricity and explain why these conditions are necessary.
The compound must be in a molten or aqueous state. In these states, the giant lattice has broken down and the ions are mobile. Mobile ions are required to act as charge carriers.
Predict the formula of the ionic compound formed between Calcium and Nitrogen.
Calcium is in Group 2 and forms a Ca(2+) ion. Nitrogen is in Group 15 and forms a N(3-) ion. To balance the charges, three Ca (2+) ions are needed for every two N(3-) The formula is:
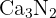
Related Articles:
Continue your A-Level Chemistry Revision with the following articles:
Summarise with AI:











