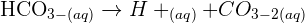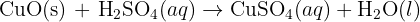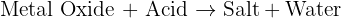Answers
Formula of Copper (II) carbonate is : CuCO3, Formula of sulfuric acid is : H2SO4CuCO3 + H2SO4 = CuSO4 + H2O + CO2
16 April 2015
The carbonate will react with the acid, the general word equation is given below:Acid + metal carbonate ---> salt + water + carbondioxideFor this reaction:copper carbonate + sulfuric acid -> copper sulfate + water + carbon dioxideTo include the oxidation numbers write: Copper (II) carbonate and copper (II) sulfate.Observations:Blue/green solid dissolves/reacts to form a blue solution; bubbles/effervescence seen as a gas is given off.
16 April 2015
Metal Carbonates generally react with Sulphuric Acid to give Metal Sulphate water and carbondioxide.
CuCO3 + H2SO4 ----------> CuSO4 + H2O + CO2
Please note the equation is already balanced.
17 April 2015
Metal carbonate will react with sulphuric acid to yield metal sulphate, water and carbon dioxide.Acid + metal carbonate ---> salt + water + carbon dioxideSo, cuprous carbonate + sulphuric acid ---> copper sulphate + water + carbon dioxideCuCO3 + H2SO4 -----> CuSO4 + H2O + CO2
31 May 2015
Metal carbonate will react with sulphuric acid to yield metal sulphate, water and carbon dioxide.Acid + metal carbonate ---> salt + water + carbon dioxideSo, cuprous carbonate + sulphuric acid ---> copper sulphate + water + carbon dioxideCuCO3 + H2SO4 -----> CuSO4 + H2O + CO2
31 May 2015
There are some basic reactions you must know in order to work out the products. Here you have a carbonate and an acid which always gives you salt,water and carbon dioxide.This is what you need to know. You need to apply this information to your reaction and then decide what salt is formed. The other two products, water and carbon dioxide remains the same for all carbonate and acid reactions.The salt formed depends on the acid used, in this case it is sulfuric acid which will form sulfates. The metal carbonate used is copper carbonate and so the salt is copper sulfate. And so your products are copper sulfate, water and carbon dioxide. If it is hot, then the reaction is going to be faster- apply collision theory which is part of the GCSE specification.
25 June 2015
5 months and no reply! Maybe there are not many tutors around... Tasnia, did you get an answer? OK This is often the example of a reaction you can do to make a soluble salt. Copper Carbonate is a green power. (usually) To make it react with sulphuric acid you need to warm the acid up but not biol it as you will get acidic fumes into your lungs. Not nice... When the mixture is heated a blue solution will start to appear. This is copper sulphate dissolved in water. (You may have seen copper sulphate as blue crystals) The short answer, products are: Copper Sulphate, Carbon Dioxide and Water.
23 September 2015
CuCO3+H2SO4~~>CuSO4+H2O+H2.
26 May 2021

Add an answer
Similar questions

 .
. 

 reacts with the carbon dioxide gas, an insoluble solid known as calcium carbonate
reacts with the carbon dioxide gas, an insoluble solid known as calcium carbonate  is generated. The equation of this reaction is given below:
is generated. The equation of this reaction is given below: