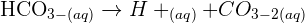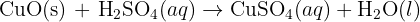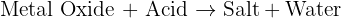moles
Assuming 65 % of a human body is water (H2O) and the rest is mostly carbon (C), estimate the number of atoms in a human body
Answers
Hi, So we can say 65 g is water which is equaivalent to 3.6 moles of water. 35 g of carbon which is equivalent to 2.9 moles of carbon. So in total there 6.5 moles in total which is equivalent to 6.5X6.023X 10^23so the answer is = 3.94 X 10^25 atoms
12 July 2016
Hi Gemma, an average human adult has a mass of around 70-80kg so lets estimate this as 75kg,65% of this is water so we have 75kgX65%=48.75kg of water, we also have 35% carbon so there is 75kgX35%=26.25kg of carbon.we can then convert these masses to number of moles using mass/molar mass=number of moles (M/n=Mol), in water the molar mass is that of 2 hydrogen atoms plus 1 oxygen atom(found from the periodic table)=1+1+16=18 and the molar mass of carbon(also form the periodic table)=12.This means we have for water 48750g/18 = 2708 molesand for carbon 26250g/12 = 2188 moles, this equals 4896 moles in total in an average humanthere are 6.022x10^23 atoms in a mole (Avagadro's constant) and so this means there are 4896x(6.022x10^23) = 2.95x10^27 atoms in an average human bodyso to estimate it is about 3x10^27 atoms :)
12 July 2016
Dear GemmaLet's consider average mass of an adult human = 70 KgTotal mass of carbon = 35x70÷100 = 24.5 KgMoles of Carbon atoms = 24.5x1000÷12 molSo , Number of Carbon atoms in a human body = 24.5x1000x 6.022 x 10^23 ÷ 12= 1.229 x 10 ^27 atoms
28 February 2019

Add an answer
Similar questions
 .
. 

 reacts with the carbon dioxide gas, an insoluble solid known as calcium carbonate
reacts with the carbon dioxide gas, an insoluble solid known as calcium carbonate  is generated. The equation of this reaction is given below:
is generated. The equation of this reaction is given below: