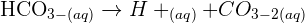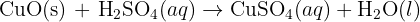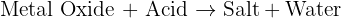increasing atomic number and reactions with oxygen and chlorine trends.
what is the trend associated by increasing atomic number and reaction with chlorine and oxygen?
Answers
I think that you need to be careful when asking this question since this is not a general trend.However if we talk about purely alkali metals, we can say this trend. This is due to the fact that as we move down the periodic table, the outer electrons have less and less attractive force to the nucleus, thus to ionically react takes less energy and so occurs more rapidly.
28 January 2015
I agree with Daniel, this is a trend seen with the alkali metals and you need to be specific in your answer, or when repeating the question...As you go down Group 1 (alkali metals) the atomic number increases, therefore there are more protons in the nucleus. Since the electrons are negatively charged there is nuclear attraction from the protons.There are always the same number of electrons as protons in neutral atoms, so the number of electrons also increases as you go down group 1. Since each electron shell can only hold so many electrons you increase the number of shells, so the outermost electron is further away from the nucleus with each descending atom, therefore the atomic radius increases (distance between outer electron and nucleus). There are more electron shells between the outer shell and nucleus, which causes electron shielding.In an A-level exam you would get 3 marks for saying, "as you go down group 1 the number of electron shells increase, therefore the atomic radius increases [1 mark]. There is also more electron shielding [1 mark] between the nucleus and outer electron, so nuclear attraction decreases [1 mark] down the group. Therefore the outer electron is more easily donated to chlorine or oxygen, and so the atom reacts more easily."Or words to that effect. But remember where you get those three marks! It's the same across exam boards and comes up practically every year!
28 January 2015
Thanks for the response.
29 January 2015
Chlorine and oxygen are both nonmetals and they react with alkali metals eg lithium and potassium to form chlorides and oxides. Points to understand include:1.Reactions between metals and non metals happen because of loss or gain of electrons. Metals react by losing electrons and non metals react by gaining electrons. To understand why, you need to be able to write the electron configurations of the metal and non metal. For chlorine it is 2,8,7, and for oxygen it is 2,6. Chlorine will react by gaining one electron to form a chloride ion, oxygen react by gaining two electrons to form an oxide ion- reason by gaining electrons they are completing their outer energy level.2. From the info in (1), you will understand that the metal will have to lose electrons for the reaction with chlorine or oxygen; Lithium has a electron configuration of 2,1 and potassium 2,8,8,1 ( higher atomic number). Which will be more reactive ie which of the two will lose the electron more easily and therefore be more reactive? It has to be potassium. The outermost electron in potassium is further away from the positive nucleus and so there is less electrostatic force between this outer electron and the positive proton; Potassium will therefore be more reactive than Lithium. And so reactivity increases with atomic number.3. The way I have explained should also help you to write the formulae of the chlorides and oxides, depending on how many electrons the metal is going to lose; Group 1 metals lose 1 outer electron, group 2 lose 2 outer electrons. I shall stop at this point because this discussion now leads on to writing formulae of ionic compounds!
29 June 2015
The answer I gave is enough for GCSE but for sixth form, we also need to use the terms jamxmitchell has used to explain the trend- more energy shells and bigger atomic radius, which means more electron shielding and so the outermost electron is less attracted to the positive nucleus and can be easily lost when compared to Lithium for example.
29 June 2015

Add an answer
Similar questions
 .
. 

 reacts with the carbon dioxide gas, an insoluble solid known as calcium carbonate
reacts with the carbon dioxide gas, an insoluble solid known as calcium carbonate  is generated. The equation of this reaction is given below:
is generated. The equation of this reaction is given below: